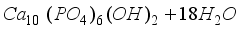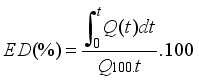Abstract
This study investigates the controlled release of insulin from composite matrices based on hydroxyapatite and Eudragit® polymers, with the aim of developing an efficient and tunable oral or implantable insulin delivery system. Hydroxyapatite was synthesized by chemical precipitation method with a Ca/P molar ratio of 1.66, closely mimicking the stoichiometry of biological apatite, and subsequently thermally treated at 800°C to enhance its crystallinity, mechanical strength, and structural stability. Two distinct matrix formulations were developed using Eudragit® RSPM and Eudragit® RS100, two pH-independent, water-insoluble polymers known for their controlled permeability properties, and were thoroughly characterized in terms of their technological and pharmacotechnical properties. Evaluation demonstrated good matrix cohesion, adequate hardness, and full compliance with mass uniformity requirements according to pharmacopoeial standards. In vitro release studies conducted under physiologically relevant conditions revealed clearly differentiated kinetic profiles depending on both the formulation type and the insulin loading dose. The first batch exhibited a classical first-order release kinetics, achieving complete insulin release over an extended period of seven days, suggesting a diffusion-controlled mechanism. In contrast, the second batch displayed a sigmoidal release profile accurately described by the Boltzmann equation (r² = 0.99), characterized by a marked acceleration phase beginning on the third day, indicative of a more complex release mechanism. These findings demonstrate that insulin loading significantly influences the release mechanism and overall kinetics, highlighting the strong potential of hydroxyapatite-polymer composite matrices for the rational design of advanced controlled insulin delivery systems.
|
Published in
|
European Journal of Biophysics (Volume 14, Issue 1)
|
|
DOI
|
10.11648/j.ejb.20261401.13
|
|
Page(s)
|
14-23 |
|
Creative Commons
|

This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited.
|
|
Copyright
|
Copyright © The Author(s), 2026. Published by Science Publishing Group
|
Keywords
Composite Matrix, Hydroxyapatite, Eudragit, Polymers, Insulin, Controlled Release
1. Introduction
An active pharmaceutical ingredient alone does not constitute a medicinal product; it generally requires an appropriate pharmaceutical carrier or delivery system to ensure effective administration within the body
| [1] | Afssaps: Liste des Excipients à Effet Notoire - Mise à jour de la liste et des libellés selon le Guideline européen 2003. (2009). 2e révision, mars 2009. |
[1]
. Medication intake and patient compliance with prescribed dosing regimens depend on several factors, among which the most common are dosing frequency
| [2] | De Vries, T. P. G., Henning, M. R. H., Hogerzeil, H. V., Fresle, D. A.: Bien prescrire les médicaments - Guide pratique.. Programme d'Action pour les Médicaments essentiels, Organisation mondiale de la Santé, Genève, Suisse. |
[2]
, treatment duration, route of administration, and the occurrence of adverse effects.
In many cases, poor treatment adherence is associated with repeated drug administration, which frequently leads to missed doses that may compromise therapeutic efficacy or delay, and in some instances negate, the intended therapeutic outcome. In this context, the development of novel pharmaceutical formulations could contribute not only to improving drug efficacy but also to enhancing patient adherence throughout the treatment period, notably by reducing dosing frequency and/or minimizing adverse effects. With respect to reducing dosing frequency, controlled-release drug delivery systems represent a particularly promising approach.
With the aim of developing devices capable of controlled drug release, we designed systems intended to ensure the sustained and continuous delivery of an active molecule, namely insulin. The objective of the present study was therefore to develop novel formulations and to evaluate the controlled release of insulin from a hydroxyapatite-based matrix. Hydroxyapatite is a ceramic material with a chemical composition similar to that of bones and teeth, which confers excellent biocompatibility and makes it a suitable excipient for the controlled delivery of active pharmaceutical substances
| [3] | Muster, D.: Biomatériaux, bio-matériels en chirurgie osseuse et dentaire (2ème partie). Encyclo. Med. Chir., Stomatologie et odontologie 22-014-F-15, 26 pages (1993). |
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
[3-5]
.
2. Materials and Methods
2.1. Laboratory Materials
The equipment used for the preparation of the delivery systems and for the investigation of drug release profiles included a porcelain mortar (EGEC 124-65), plastic molds with a diameter of 12 mm (Aventis Pharma, Dakar, Senegal), 10 mL and 100 mL Pyrex glass graduated cylinders (Prolabo, Paris, France), graduated pipettes of 1, 5, and 10 mL (Suprema, Prolabo, Paris, France), a 500 µL Eppendorf precision pipette, adjustable Finnpipette micropipettes (50, 100, 150, and 200 µL), a Mettler H16 analytical balance, a D135-I drying oven (Kowel), Pyrex volumetric flasks of 50, 100, 250, 500, and 1000 mL (Prolabo, Paris, France), a mercury thermometer (−40 to 250°C) (Prolabo, Paris, France), a heated magnetic stirrer (Fisher Scientific, USA), Pyrex Erlenmeyer flasks (Prolabo, Paris, France), a 50/0.1 cm³ Aspin burette, a Volca 33-55 muffle furnace (Prolabo, Paris, France), a UV-visible spectrophotometer (Jenway 6300), Pyrex vacuum flasks (Prolabo, Paris, France), test tubes (16 × 150 mm) (Deltalabo, Barcelona, Spain), alumina crucibles (Prolabo, Paris, France), porcelain Büchner funnels (Prolabo, Paris, France), and Whatman filter papers.
The insulin used in this study was Actrapid® HM, a human insulin produced by Novo Nordisk using recombinant DNA (rDNA) technology in Saccharomyces cerevisiae. Following subcutaneous administration, its maximum plasma concentration is typically reached between 1.5 and 2.5 hours. Actrapid® HM is indicated for the treatment of type I diabetes mellitus.
Hydroxyapatite powder was used as the main excipient for the development of the controlled-release systems. The polymers Eudragit® L100 and Eudragit® RS PM were also employed in the formulation of the matrices.
2.2. Methods
2.2.1. Preparation of Hydroxyapatite
Hydroxyapatite was prepared by direct precipitation by adding, under continuous stirring, a 0.3 M phosphoric acid (H
3PO
4) solution at a flow rate of 25 mL·min⁻
1 to a freshly prepared 0.5 M calcium hydroxide (Ca(OH)₂) suspension, according to the following reaction
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
| [6] | Marchat, D., Zymelka, M., Coelho, C., Gremillard, L., Joly-Pottuz, L., Babonneau, F., Esnouf, C., Chevalier, J., Bernache-Assollant, D.: Accurate characterization of pure silicon-substituted hydroxyapatite powders synthesized by a new precipitation route. Acta Biomaterialia 9(6), 6992-7004 (2013).
https://doi.org/10.1016/j.actbio.2013.03.011 |
| [7] | Bang, L. T., Long, B. D., Othman, R.: Carbonate hydroxyapatite and silicon-substituted carbonate hydroxyapatite: synthesis, mechanical properties, and solubility evaluations. The Scientific World Journal 2014, 969876 (2014).
https://doi.org/10.1155/2014/969876 |
[4-7]
:
6H3PO4+ 10Ca (OH)2󠄀Ca10(PO4)6(OH)2+ 18H2O
The Ca/P molar ratio was fixed at 1.66. The precipitation reaction was carried out under continuous stirring, either at 20°C (sample no. 1) or at a temperature ranging between 80 and 95°C (sample no. 2).
When precipitation was performed at 20°C, stirring was maintained for 45 min after completion of phosphoric acid addition. The resulting suspension was then allowed to age for 24 h. The precipitate was recovered by filtration and dried in an oven with a gradual temperature increase from 40 to 80°C over 24 h, followed by a holding period of 5 h at 80°C. Heating the precipitate at 80°C allowed the formation of stoichiometric hydroxyapatite
| [6] | Marchat, D., Zymelka, M., Coelho, C., Gremillard, L., Joly-Pottuz, L., Babonneau, F., Esnouf, C., Chevalier, J., Bernache-Assollant, D.: Accurate characterization of pure silicon-substituted hydroxyapatite powders synthesized by a new precipitation route. Acta Biomaterialia 9(6), 6992-7004 (2013).
https://doi.org/10.1016/j.actbio.2013.03.011 |
| [7] | Bang, L. T., Long, B. D., Othman, R.: Carbonate hydroxyapatite and silicon-substituted carbonate hydroxyapatite: synthesis, mechanical properties, and solubility evaluations. The Scientific World Journal 2014, 969876 (2014).
https://doi.org/10.1155/2014/969876 |
| [8] | Panda, S., Biswas, C. K., Paul, S.: A comprehensive review on the preparation and application of calcium hydroxyapatite: A special focus on atomic doping methods for bone tissue engineering. Ceramics International 47(20), 28122-28144 (2021).
https://doi.org/10.1016/j.ceramint.2021.07.100 |
[6-8]
.
When precipitation was carried out at elevated temperatures (80-95°C), both stirring and heating were maintained for 20 min, after which heating was stopped while stirring was continued for an additional 25 min. The suspension was then aged for 24 h, and the subsequent steps (filtration and drying) were identical to those described above.
The obtained hydroxyapatite powder samples were subsequently calcined at 600°C, 800°C, 1000°C, and 1200°C for 3 h, with a heating rate of 4°C·min⁻
1 (
Table 2), in order to promote hydroxyapatite crystallization and to obtain powders with different granulometries
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
[4, 5]
.
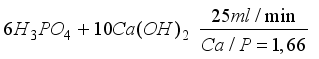
>
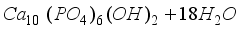
2.2.2. Characterization
X-ray diffraction (XRD)
The samples were characterized by X-ray diffraction using a D500 diffractometer (Siemens, Karlsruhe, Germany) equipped with a front quartz crystal monochromator (Co Kα₁ = 0.178897 nm). The characteristic diffraction peaks of hydroxyapatite (JCPDS file No. 9-432) were observed in the XRD patterns of all samples.
However, the diffraction patterns of sample 2 exhibited significantly sharper and better-defined peaks compared to those of sample 1, indicating a higher degree of crystallinity. In addition, the continuous background was lower for sample 2, suggesting a reduced proportion of amorphous phase relative to sample 1. Furthermore, diffraction peaks corresponding to tricalcium phosphate Ca
3(PO
4)₂ (α-TCP) were absent after calcination in sample 2, whereas they were still detected in sample 1 (
Table 1)
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [9] | Chafik El Idrissi, B.: A novel method to synthesize nanocrystalline hydroxyapatite: Characterization with x-ray diffraction and infrared spectroscopy. IOSR Journal of Applied Chemistry 7(5), 107-112 (2014).
https://doi.org/10.9790/5736-0753107112 |
| [10] | Kaygili, O., Dorozhkin, S. V., Keser, S.: Synthesis and characterization of Ce-substituted hydroxyapatite by sol-gel method. Materials Science and Engineering: C 42, 78-82 (2014). https://doi.org/10.1016/j.msec.2014.05.024 |
| [11] | Lin, Y., Yang, Z., Cheng, J.: Preparation, Characterization and Antibacterial Property of Cerium Substituted Hydroxyapatite Nanoparticles. Journal of Rare Earths 25(4), 452-456 (2007).
https://doi.org/10.1016/S1002-0721(07)60455-4 |
[4, 9-11]
.
The hydroxyapatite obtained after calcination of sample 2 at 600°C, 800°C, 1000°C, and 1200°C was well crystallized and free of detectable impurities (
Figure 1). Consequently, this sample was selected for the development of controlled-release systems. For this reason, all subsequent characterizations were performed exclusively on sample 2.
Table 1.
Crystalline phases identified by X-ray diffraction as a function of precipitation temperature and maturation time, after a 3-hour dwell at different calcination temperatures | [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
Sample | Precipitation temperature (°C) | Maturation time (h) | 80 °C | 600 °C | 800 °C | 1000 °C | 1200 °C |
Sample 1 | 20 | 24 | Apatite + amorphous phase | Apatite + amorphous phase | Apatite | Apatite + -TCP | Apatite + -TCP |
Sample 2 | 80-95 | 24 | Apatite + amorphous phase | Apatite | Apatite | Apatite | Apatite |
Figure 1. X-ray diffraction patterns of thermally treated hydroxyapatite (Sample 2).
XRD patterns of hydroxyapatite precipitated at 80-95°C and dried at 80°C (a), after calcination in air for 3 h at 600°C (b), 800°C (c), 1000°C (d), and 1200°C (e), showing progressive improvement in crystallinity
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
[4]
.
Infrared Spectroscopy (FTIR)
FTIR spectra of Sample 2 before and after calcination were obtained using an ATI Mattson Genesis Series FTIR™ spectrophotometer (GMI, Strasbourg, France). All spectra closely matched that of standard synthetic hydroxyapatite (
Figure 2), confirming the preservation of the apatite structure after thermal treatment. The presence of hydroxyl groups was evidenced by absorption bands at 3571 and 630 cm⁻
1, whereas the characteristic phosphate (PO
4³⁻) vibrational modes appeared at 571, 601, 962, and 1040-1080 cm⁻
1 | [10] | Kaygili, O., Dorozhkin, S. V., Keser, S.: Synthesis and characterization of Ce-substituted hydroxyapatite by sol-gel method. Materials Science and Engineering: C 42, 78-82 (2014). https://doi.org/10.1016/j.msec.2014.05.024 |
| [11] | Lin, Y., Yang, Z., Cheng, J.: Preparation, Characterization and Antibacterial Property of Cerium Substituted Hydroxyapatite Nanoparticles. Journal of Rare Earths 25(4), 452-456 (2007).
https://doi.org/10.1016/S1002-0721(07)60455-4 |
[10, 11]
.
Specific surface area of powders
The specific surface area of Sample 2 and of the powders obtained after thermal treatment of Sample 2 (
Table 1) was determined by nitrogen adsorption using the BET method with a Sorpty 1750 apparatus (Carlo Erba Strumentazione, Milan, Italy). The results show that the specific surface area increases with increasing calcination temperature up to 800°C, beyond which a marked decrease is observed.
Table 2. Variation of the specific surface area of the powder precipitated at 80-95°C as a function of thermal treatment temperature (BET method).
Annealing temperature (°C) | 80 | 600 | 800 | 1000 | 1200 |
BET surface area (m2/g) | 40 | 50 | 75 | 7 | 5 |
Selected Powder Samples
The powder selected for the preparation of the hydroxyapatite-polymer composite system intended for the controlled release of insulin was calcined hydroxyapatite (CHA). This material was obtained from non-calcined hydroxyapatite, which was first filtered and dried in an oven at 80°C for 24 h, followed by calcination at 800°C for 3 h.
The resulting calcined hydroxyapatite exhibited a specific surface area of 75 m2/g and consisted of particles with sizes ranging from 100 to 200 nm. Prior to use, all powder samples were sieved through a mesh with a nominal aperture size of 250 µm to ensure particle size homogeneity.
Preparation of the Controlled Release Systems
The controlled release of active pharmaceutical ingredients from hydroxyapatite-based matrices relies on the ability of this material to adsorb and retain bioactive compounds. Numerous studies have demonstrated the relevance of hydroxyapatite for the development of controlled release systems, either in the form of tablets intended for oral or buccal administration or as cement-based systems for bone-related applications
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [12] | Kareem, R. O., Bulut, N., Kaygili, O.: Hydroxyapatite Biomaterials: A Comprehensive Review of their Properties, Structures, Medical Applications, and Fabrication Methods. Journal of Chemical Reviews 6(1), 1-26 (2024).
https://doi.org/10.48309/jcr.2024.415051.1253 |
| [13] | Szcześ, A., Hołysz, L., Chibowski, E.: Synthesis of hydroxyapatite for biomedical applications. Advances in Colloid and Interface Science 249, 321-330 (2017).
https://doi.org/10.1016/j.cis.2017.04.007 |
| [14] | Akram, M., Alshemary, A. Z., Goh, Y.-F., Ibrahim, W. A. W., Lintang, H. O., Hussain, R.: Continuous Microwave Flow Synthesis of Mesoporous Hydroxyapatite. Materials Science & Engineering C 56, 356-362 (2015).
https://doi.org/10.1016/j.msec.2015.06.040 |
| [15] | Basu, P.: In situ synthesis of hydroxyapatite/carboxymethyl cellulose composites for bone regeneration applications. Colloid and Polymer Science 296(10), 1729-1738 (2018).
https://doi.org/10.1007/S00396-018-4393-9 |
| [16] | Sivakumar, M., Manjubala, I., Panduranga Rao, K.: Preparation, characterization and in-vitro release of gentamicin from coralline hydroxyapatite-chitosan composite microspheres. Carbohydrate Polymers 49(3), 281-288 (2002).
https://doi.org/10.1016/S0144-8617(01)00331-9 |
| [17] | Bodylska, W., Junka, A., Brożyna, M., Bartmański, M., Gadzała-Kopciuch, R., Jaromin, A., Navarro, J. A. R., Lukowiak, A., Fandzloch, M.: New Biocompatible Ti-MOF@hydroxyapatite Composite Boosted with Gentamicin for Postoperative Infection Control. ACS Biomaterials Science & Engineering 10(12), 7555-7565 (2024).
https://doi.org/10.1021/acsbiomaterials.4c01230 |
| [18] | Munir, M. U., Salman, S., Javed, I., Bukhari, S. N. A., Ahmad, N., Shad, N. A., Aziz, F.: Nano-hydroxyapatite as a delivery system: overview and advancements. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 717-727 (2021).
https://doi.org/10.1080/21691401.2021.2016785 |
[4, 12-18]
.
In this context, insulin-controlled release systems were developed using calcined hydroxyapatite in combination with Eudragit®-type polymers. The resulting formulations consisted of hydroxyapatite, the active ingredient (insulin), and polymeric binders, namely Eudragit® RS PM and Eudragit® RL 100, the latter being used as an external-phase binder.
The manufacturing process was based on the prior adsorption of insulin onto hydroxyapatite, followed by homogeneous mixing with the selected polymers. The resulting mixtures were then molded into plastic cavities and dried at room temperature. This protocol enabled the preparation of two distinct batches, each consisting of ten hydroxyapatite-polymer-drug systems intended for the evaluation of insulin-controlled release behavior.
The composition of the different batches is summarized in
Table 3.
Table 3. Composition of the different hydroxyapatite-polymer-insulin systems expressed as weight percentages.
Batch | Annealed HAp (%) | Eudragit® internal (%) | Eudragit® external (%) | Insulin (%) |
Batch 1 | 76.79 | 15.38 | 7.7 | 0.135 |
Batch 2 | 76.65 | 15.38 | 7.7 | 0.270 |
For each batch, ten units were prepared; the molds used (blister cavities) had a diameter of 12 mm and a depth of 4 mm (
Figure 3).
Figure 3. Schematic representation of the homogeneous systems.
All the prepared systems were weighed after oven drying, allowing the determination of the mean mass, standard deviation, as well as the minimum and maximum masses.
3. Results
3.1. Evaluation of the Systems
Mass uniformity test
All the prepared systems were weighed after drying at ambient temperature. The mean mass, standard deviation, as well as the minimum and maximum masses permitted according to pharmacopoeial requirements were then calculated.
All the studied batches were found to be compliant; indeed, we observed that:
The results of these calculations are reported in
Table 4.
Table 4. Pharmacotechnical parameters of the systems.
Batch | Mean mass (g) | Standard deviation (g) | Authorized maximum (g) | Authorized minimum (g) |
Lot 1 | 0.103 | 1.99.10-5 | 0.10815 | 0.09785 |
Lot 2 | 0.104 | 3.95.10-5 | 0.1092 | 0.0988 |
Study of insulin release
The insulin release study was carried out in an aqueous medium. For each batch, the experiment was performed on three units of known mass. Insulin concentrations were determined using the Biuret method.
The mean cumulative release profile was calculated from a calibration curve, as shown in
Figure 4:
Figure 4. Calibration curve.
Insulin release was evaluated in vitro using a standardized protocol. The experiments were carried out in cylindrical plastic vessels with a capacity of 150 mL, containing 100 mL of distilled water and sealed with a screw cap to minimize evaporation. Representative samples from each batch were immersed in the dissolution medium without agitation and maintained at room temperature.
Aliquots of 2 mL were withdrawn every 24 h, and each sampled volume was immediately replaced with an equal volume of distilled water after homogenization of the medium by gentle inversion of the vessel. The collected samples were mixed with 5 mL of Biuret reagent, and the amount of insulin released was quantified by measuring the optical density at 550 nm using a UV-visible spectrophotometer (Jenway, model 6300).
3.2. Release Profiles of the Systems
Since ceramic-based controlled release systems do not release their entire active substance load within 12 hours, sampling was performed every 24 hours. The release studies conducted on the different batches were carried out over a period of 7 days. The results are presented in the form of curves and tables. The amounts of insulin released, expressed as milligrams per unit surface area Q (mg/cm2), as a function of time for the samples from batch 1 are shown in the following figure:
Figure 5. Insulin release profile of batch 1 expressed as mg/cm2.
From analysis of the release curves, a first-order kinetic release profile can be identified. This behavior can be described by the following relationship:
Q is the amount of insulin released per unit surface area at time ttt;
y0 is the amount of insulin released per unit surface area after seven days;
A1 is a dimensionless constant;
t is the characteristic time constant of the release process
For both batches, a higher dissolution efficiency was observed at lower insulin loadings, except during the initial phase up to day 3, where this trend was not clearly established. By examining the shape of the release curves for batch 2, it can be observed that the release profiles can be modeled using the Boltzmann equation, which describes a sigmoidal behavior. This relationship is expressed by the following equation:
Q represents the amount of insulin released;
A1 and A2A are release constants corresponding to the lower and upper asymptotes of the sigmoidal curve;
x0 is the time corresponding to the inflection point of the release curve;
dx is the characteristic time constant related to the slope of the release profile.
The amounts released, expressed as milligrams per unit surface area Qi (mg/cm
2), as a function of time for batch 2 are shown in the following figure. The modeling parameters as well as the correlation coefficient were calculated using Origin® software (version 6.1) and are also presented in
Figure 7.
Figure 6. Insulin release profile of batch 2 expressed as mg/cm2.
In
Figures 7 and 8, the percentages of insulin released Qi (%) from the matrices are presented. It can be observed that the batches of systems containing the lowest insulin concentrations exhibit faster release compared with batches formulated with higher insulin loadings.
Figure 7. Percentage of insulin released from batch 1.
Figure 8. Percentage of insulin released from batch 2.
The mean insulin dissolution efficiencies for both batches were calculated. This parameter was proposed by Khan
| [20] | Li, D., Huang, X., Wu, Y., Li, J., Cheng, W., He, J., Tian, H., Huang, Y.: Preparation of pH-responsive mesoporous hydroxyapatite nanoparticles for intracellular controlled release of an anticancer drug. Biomaterials Science 4, 272-280 (2016).
https://doi.org/10.1039/C5BM00228A |
[20]
and is defined as the ratio between the area under the dissolution curve up to time ttt and the area of the rectangle corresponding to 100% dissolution at the same time. For comparative purposes, identical and fixed time intervals must be selected.
ED (%) is the dissolution efficiency at time ttt;
Q(t) is the amount of active pharmaceutical ingredient released as a function of time;
Q100 is the total amount of active ingredient contained in the tablet.
Table 5. Mean dissolution efficiencies of the systems.
Time (days) | Cumulative SQ1 released (%) |
3 | 10.61 ± 0.49 |
5 | 33.57 ± 1.46 |
7 | 61.95 ± 1.63 |
For both batches, the dissolution efficiency was found to be higher when the insulin loading was lower, except during the initial period ending on day 3.
4. Discussion
The continuous release of active pharmaceutical ingredients is currently a major focus in the pharmaceutical industry; therefore, this study investigated the release of insulin from hydroxyapatite-based matrices.
The choice of hydroxyapatite as the main excipient is justified by its mineral composition, which is close to that of bone and dental tissues, as well as by its proven suitability for the development of sustained release systems
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
| [12] | Kareem, R. O., Bulut, N., Kaygili, O.: Hydroxyapatite Biomaterials: A Comprehensive Review of their Properties, Structures, Medical Applications, and Fabrication Methods. Journal of Chemical Reviews 6(1), 1-26 (2024).
https://doi.org/10.48309/jcr.2024.415051.1253 |
| [18] | Munir, M. U., Salman, S., Javed, I., Bukhari, S. N. A., Ahmad, N., Shad, N. A., Aziz, F.: Nano-hydroxyapatite as a delivery system: overview and advancements. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 717-727 (2021).
https://doi.org/10.1080/21691401.2021.2016785 |
[4, 5, 12, 18]
. The hydroxyapatite powder used in this work was obtained by a precipitation reaction between calcium hydroxide and orthophosphoric acid with a Ca/P ratio of 1.66, carried out at temperatures ranging between 80 and 95°C. After filtration and drying at 80°C for 24 h, the powder was calcined at 800°C for 3 h.
In addition, polymeric excipients were incorporated into the formulations, namely Eudragit® RS PM in powder form as the internal phase and Eudragit® RS100 as the external phase using absolute ethanol as the wetting liquid. Two batches of composite systems were obtained, whose individual units exhibited good mechanical consistency. Mass uniformity testing demonstrated that all systems complied with pharmacopoeial requirements.
Insulin release studies performed in an aqueous medium revealed that the release profiles followed different kinetic laws depending on the matrix composition. For batch 1, insulin release followed a first-order kinetic profile, modeled by the equation Q=y0 + A1e-x/t.
These release profiles are similar to those previously obtained with paracetamol, for which release extended over 18 days
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
[5]
, whereas insulin release was completed within 7 days. This difference can be attributed to the initial drug loading. In insulin-based systems, insulin concentrations ranged from 0.135% to 0.27%, whereas paracetamol loadings reported in the literature vary between 5% and 30%
| [4] | Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg. |
| [5] | Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009). |
| [17] | Bodylska, W., Junka, A., Brożyna, M., Bartmański, M., Gadzała-Kopciuch, R., Jaromin, A., Navarro, J. A. R., Lukowiak, A., Fandzloch, M.: New Biocompatible Ti-MOF@hydroxyapatite Composite Boosted with Gentamicin for Postoperative Infection Control. ACS Biomaterials Science & Engineering 10(12), 7555-7565 (2024).
https://doi.org/10.1021/acsbiomaterials.4c01230 |
| [18] | Munir, M. U., Salman, S., Javed, I., Bukhari, S. N. A., Ahmad, N., Shad, N. A., Aziz, F.: Nano-hydroxyapatite as a delivery system: overview and advancements. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 717-727 (2021).
https://doi.org/10.1080/21691401.2021.2016785 |
[4, 5, 17, 18]
.
For batch 2, the release kinetics were best described by the Boltzmann equation, yielding a sigmoidal release profile. The selection of the Boltzmann model was justified by its higher correlation coefficient (r
2 = 0.99) compared with that obtained using first-order kinetics (r
2 = 0.96). In fact, batch 2 exhibited a relatively sharp increase in insulin release around the third day, suggesting a release mechanism that may be specific to insulin and, more generally, to protein-based active substances
| [19] | Munir, M. U., Ihsan, A., Javed, I., Ansari, M. T., Bajwa, S. Z., Bukhari, S. N. A., Ahmed, A., Malik, M. Z., Khan, W. S.: Controllably Biodegradable Hydroxyapatite Nanostructures for Cefazolin Delivery against Antibacterial Resistance. ACS Omega 4(4), 7524-7532 (2019).
https://doi.org/10.1021/acsomega.9b00541 |
| [20] | Li, D., Huang, X., Wu, Y., Li, J., Cheng, W., He, J., Tian, H., Huang, Y.: Preparation of pH-responsive mesoporous hydroxyapatite nanoparticles for intracellular controlled release of an anticancer drug. Biomaterials Science 4, 272-280 (2016).
https://doi.org/10.1039/C5BM00228A |
| [21] | Palard, M., Combes, J., Champion, E., Foucaud, S., Rattner, A., Bernache-Assollant, D.: Effect of silicon content on the sintering and biological behaviour of Ca10(PO4)(6-x)(SiO4)x(OH)(2-x) ceramics. Acta Biomaterialia 5(4), 1223-1232 (2009).
https://doi.org/10.1016/j.actbio.2008.10.016 |
| [22] | Lytkina, D. N., Fedorishin, D. A., Kalachikova, P. M., Plyaskina, A. A., Babeshin, A. R., Kurzina, I. A.: Cryo-Structured Materials Based on Polyvinyl Alcohol and Hydroxyapatite for Osteogenesis. Journal of Functional Biomaterials 12(1), 18 (2021).
https://doi.org/10.3390/jfb12010018 |
| [23] | Alizadeh-Osgouei, M., Li, Y., Wen, C.: A comprehensive review of biodegradable synthetic polymer-ceramic composites and their manufacture for biomedical applications. Bioactive Materials 4(1), 22-36 (2019).
https://doi.org/10.1016/j.bioactmat.2018.11.003 |
| [24] | Jiménez-Pérez, A., Martínez-Alonso, M., García-Tojal, J.: Hybrid Hydroxyapatite-Metal Complex Materials Derived from Amino Acids and Nucleobases. Molecules 29(18), 4479 (2024).
https://doi.org/10.3390/molecules29184479 |
[19-24]
.
The analysis of dissolution efficiencies, which increased as insulin loading decreased, further confirmed that batch 1 released the entire insulin content by the third day, whereas batch 2 released only approximately 60% of its initial insulin load over the same period. These findings demonstrate that the initial insulin content plays a key role in determining the release profile of insulin from hydroxyapatite-polymer matrices
| [18] | Munir, M. U., Salman, S., Javed, I., Bukhari, S. N. A., Ahmad, N., Shad, N. A., Aziz, F.: Nano-hydroxyapatite as a delivery system: overview and advancements. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 717-727 (2021).
https://doi.org/10.1080/21691401.2021.2016785 |
| [25] | Predoi, D., Ciobanu, S. C., Iconaru, S. L., Ţălu, Ş., Ghegoiu, L., Matos, R. S., da Fonseca Filho, H. D., Trusca, R.: New Physico-Chemical Analysis of Magnesium-Doped Hydroxyapatite in Dextran Matrix Nanocomposites. Polymers 16(1), 125 (2024). https://doi.org/10.3390/polym16010125 |
| [26] | Geuli, O., Metoki, N., Zada, T., Reches, M., Eliaz, N., Mandler, D.: Synthesis, coating, and drug-release of hydroxyapatite nanoparticles loaded with antibiotics. Journal of Materials Chemistry B 5, 7819-7830 (2017).
https://doi.org/10.1039/C7TB02105D |
| [27] | Chung, J. H., Kim, Y. K., Kim, K. H., Kwon, T. Y., Vaezmomeni, S. Z., Samiei, M., Akbarzadeh, A.: Synthesis, characterization, biocompatibility of hydroxyapatite-natural polymers nanocomposites for dentistry applications. Artificial Cells, Nanomedicine, and Biotechnology 44(1), 277-284 (2016).
https://doi.org/10.3109/21691401.2014.944644 |
| [28] | Hassanzadeh, A., Ashrafihelan, J., Salehi, R., Rahbarghazi, R., Firouzamandi, M., Ahmadi, M., Aghazadeh, M.: Development and biocompatibility of the injectable collagen/nano-hydroxyapatite scaffolds as in situ forming hydrogel for the hard tissue engineering application. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 136-146 (2021).
https://doi.org/10.1080/21691401.2021.1877153 |
[18, 25-28]
.
Overall, comparison of the release profiles of the two batches showed no major difference between them. The statistical significance of the observed differences was evaluated using Student’s t-test for small independent samples, yielding a p-value of approximately 0.76, which is higher than the conventional threshold of 0.05 and also greater than the threshold recommended by Ronald Fisher. According to Fisher, a p-value greater than 0.1 indicates no evidence against the null hypothesis.
5. Conclusion
At present, one of the major challenges faced by the pharmaceutical industry is the development of new dosage forms based on existing active pharmaceutical ingredients in order to improve their therapeutic effectiveness after administration. In this context, the present work focused on the study of insulin release from a hydroxyapatite-based matrix with the aim of defining and understanding its release profile. Hydroxyapatite was selected as the main excipient. The hydroxyapatite powder used in this study was obtained by a precipitation reaction between calcium hydroxide and orthophosphoric acid with a Ca/P ratio of 1.66, carried out at temperatures ranging from 80 to 95°C. After filtration and drying at 80°C for 24 h, the powder was calcined at 800°C for 3 h. Polymeric excipients were also incorporated into the formulations, namely Eudragit® RS PM in powder form as the internal phase and Eudragit® RS100 as the external phase using absolute ethanol as a wetting agent. Two batches of composite systems were obtained, whose individual units exhibited good mechanical integrity. Mass uniformity testing demonstrated that all systems complied with pharmacopoeial requirements. Insulin release studies performed in aqueous medium showed that the release profiles followed different kinetic behaviors depending on the matrix composition. For batch 1, insulin release followed a first-order kinetic profile modeled by the equation Q=y0 + A1e-x/t.with complete release achieved within 7 days. This relatively short release duration was directly related to the initial insulin loading in the system.
For batch 2, the release kinetics were best described by the Boltzmann equation, leading to a sigmoidal release profile. The use of the Boltzmann model was justified by a higher correlation coefficient (r2 = 0.99) compared with that obtained using first-order kinetics (r2 = 0.96). In this batch, a relatively abrupt increase in insulin release was observed around the third day, suggesting a release behavior that may be specific to insulin and, more generally, to protein-based active substances. The analysis of dissolution efficiencies, which increased as insulin loading decreased, confirmed that batch 1 released a large proportion of insulin from the third day, whereas batch 2 released only about 60% of its initial insulin content over the same period. These results highlight the crucial influence of the initial insulin loading on the release behavior. Overall, the findings of this study demonstrate that hydroxyapatite powder constitutes a suitable substrate for the development of hydroxyapatite-polymer matrices capable of achieving controlled and prolonged insulin release. Nevertheless, the kinetic profile observed for batch 2 requires further investigation, as the discontinuous release pattern suggests a concentration-dependent behavior that may indicate a two-phase release mechanism, characterized by an initial burst effect followed by a more gradual elution phase.
Abbreviations
XRD | X-ray Diffraction |
FTIR | Fourier Transform Infrared Spectroscopy |
BET | Brunauer Emmett Teller |
Author Contributions
Abdou Faye: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing
Sidy Mouhamed Dieng: Supervision, Data curation, Investigation, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing
Marie Jeanne Arlette Ciss: Validation, Visualization, Writing – review & editing
Alphonse Rodrigue Djiboune: Validation, Visualization, Writing – review & editing
Benedicte Souho: Validation, Visualization, Writing – review & editing
Mamadou Soumboundou: Validation, Visualization, Writing – review & editing
Papa Mady Sy: Validation, Visualization, Writing – review & editing
Gora Mbaye: Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing
Mounibe Diarra: Supervision, Validation, Visualization,
Writing – original draft, Writing – review & editing
Louis Augustin Diaga Diouf: Conceptualization, Formal Analysis, Investigation, Methodology, Resources, Supervision, Validation, Visualization, Writing – review & editing
Conflicts of Interest
The authors declare no conflicts of interest.
References
| [1] |
Afssaps: Liste des Excipients à Effet Notoire - Mise à jour de la liste et des libellés selon le Guideline européen 2003. (2009). 2e révision, mars 2009.
|
| [2] |
De Vries, T. P. G., Henning, M. R. H., Hogerzeil, H. V., Fresle, D. A.: Bien prescrire les médicaments - Guide pratique.. Programme d'Action pour les Médicaments essentiels, Organisation mondiale de la Santé, Genève, Suisse.
|
| [3] |
Muster, D.: Biomatériaux, bio-matériels en chirurgie osseuse et dentaire (2ème partie). Encyclo. Med. Chir., Stomatologie et odontologie 22-014-F-15, 26 pages (1993).
|
| [4] |
Diarra, M., Pourroy, G., Boymond, C., Muster, D.: Elaboration et évaluation d'un système hydroxyapatite/polymère pour une libération contrôlée de substances actives au niveau de la cavité buccale.. Thèse de troisième cycle n°3247, Université Louis Pasteur, Strasbourg.
|
| [5] |
Diouf, L. A. D., Mbaye, G., Ndiaye, A., Diedhiou, A., Cissé, A., & Diarra, M. Développement d’un système hydroxyapatite/polymères pour la libération contrôlée du paracétamol. Dakar Médical, 54(1), 14–22 (2009).
|
| [6] |
Marchat, D., Zymelka, M., Coelho, C., Gremillard, L., Joly-Pottuz, L., Babonneau, F., Esnouf, C., Chevalier, J., Bernache-Assollant, D.: Accurate characterization of pure silicon-substituted hydroxyapatite powders synthesized by a new precipitation route. Acta Biomaterialia 9(6), 6992-7004 (2013).
https://doi.org/10.1016/j.actbio.2013.03.011
|
| [7] |
Bang, L. T., Long, B. D., Othman, R.: Carbonate hydroxyapatite and silicon-substituted carbonate hydroxyapatite: synthesis, mechanical properties, and solubility evaluations. The Scientific World Journal 2014, 969876 (2014).
https://doi.org/10.1155/2014/969876
|
| [8] |
Panda, S., Biswas, C. K., Paul, S.: A comprehensive review on the preparation and application of calcium hydroxyapatite: A special focus on atomic doping methods for bone tissue engineering. Ceramics International 47(20), 28122-28144 (2021).
https://doi.org/10.1016/j.ceramint.2021.07.100
|
| [9] |
Chafik El Idrissi, B.: A novel method to synthesize nanocrystalline hydroxyapatite: Characterization with x-ray diffraction and infrared spectroscopy. IOSR Journal of Applied Chemistry 7(5), 107-112 (2014).
https://doi.org/10.9790/5736-0753107112
|
| [10] |
Kaygili, O., Dorozhkin, S. V., Keser, S.: Synthesis and characterization of Ce-substituted hydroxyapatite by sol-gel method. Materials Science and Engineering: C 42, 78-82 (2014).
https://doi.org/10.1016/j.msec.2014.05.024
|
| [11] |
Lin, Y., Yang, Z., Cheng, J.: Preparation, Characterization and Antibacterial Property of Cerium Substituted Hydroxyapatite Nanoparticles. Journal of Rare Earths 25(4), 452-456 (2007).
https://doi.org/10.1016/S1002-0721(07)60455-4
|
| [12] |
Kareem, R. O., Bulut, N., Kaygili, O.: Hydroxyapatite Biomaterials: A Comprehensive Review of their Properties, Structures, Medical Applications, and Fabrication Methods. Journal of Chemical Reviews 6(1), 1-26 (2024).
https://doi.org/10.48309/jcr.2024.415051.1253
|
| [13] |
Szcześ, A., Hołysz, L., Chibowski, E.: Synthesis of hydroxyapatite for biomedical applications. Advances in Colloid and Interface Science 249, 321-330 (2017).
https://doi.org/10.1016/j.cis.2017.04.007
|
| [14] |
Akram, M., Alshemary, A. Z., Goh, Y.-F., Ibrahim, W. A. W., Lintang, H. O., Hussain, R.: Continuous Microwave Flow Synthesis of Mesoporous Hydroxyapatite. Materials Science & Engineering C 56, 356-362 (2015).
https://doi.org/10.1016/j.msec.2015.06.040
|
| [15] |
Basu, P.: In situ synthesis of hydroxyapatite/carboxymethyl cellulose composites for bone regeneration applications. Colloid and Polymer Science 296(10), 1729-1738 (2018).
https://doi.org/10.1007/S00396-018-4393-9
|
| [16] |
Sivakumar, M., Manjubala, I., Panduranga Rao, K.: Preparation, characterization and in-vitro release of gentamicin from coralline hydroxyapatite-chitosan composite microspheres. Carbohydrate Polymers 49(3), 281-288 (2002).
https://doi.org/10.1016/S0144-8617(01)00331-9
|
| [17] |
Bodylska, W., Junka, A., Brożyna, M., Bartmański, M., Gadzała-Kopciuch, R., Jaromin, A., Navarro, J. A. R., Lukowiak, A., Fandzloch, M.: New Biocompatible Ti-MOF@hydroxyapatite Composite Boosted with Gentamicin for Postoperative Infection Control. ACS Biomaterials Science & Engineering 10(12), 7555-7565 (2024).
https://doi.org/10.1021/acsbiomaterials.4c01230
|
| [18] |
Munir, M. U., Salman, S., Javed, I., Bukhari, S. N. A., Ahmad, N., Shad, N. A., Aziz, F.: Nano-hydroxyapatite as a delivery system: overview and advancements. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 717-727 (2021).
https://doi.org/10.1080/21691401.2021.2016785
|
| [19] |
Munir, M. U., Ihsan, A., Javed, I., Ansari, M. T., Bajwa, S. Z., Bukhari, S. N. A., Ahmed, A., Malik, M. Z., Khan, W. S.: Controllably Biodegradable Hydroxyapatite Nanostructures for Cefazolin Delivery against Antibacterial Resistance. ACS Omega 4(4), 7524-7532 (2019).
https://doi.org/10.1021/acsomega.9b00541
|
| [20] |
Li, D., Huang, X., Wu, Y., Li, J., Cheng, W., He, J., Tian, H., Huang, Y.: Preparation of pH-responsive mesoporous hydroxyapatite nanoparticles for intracellular controlled release of an anticancer drug. Biomaterials Science 4, 272-280 (2016).
https://doi.org/10.1039/C5BM00228A
|
| [21] |
Palard, M., Combes, J., Champion, E., Foucaud, S., Rattner, A., Bernache-Assollant, D.: Effect of silicon content on the sintering and biological behaviour of Ca10(PO4)(6-x)(SiO4)x(OH)(2-x) ceramics. Acta Biomaterialia 5(4), 1223-1232 (2009).
https://doi.org/10.1016/j.actbio.2008.10.016
|
| [22] |
Lytkina, D. N., Fedorishin, D. A., Kalachikova, P. M., Plyaskina, A. A., Babeshin, A. R., Kurzina, I. A.: Cryo-Structured Materials Based on Polyvinyl Alcohol and Hydroxyapatite for Osteogenesis. Journal of Functional Biomaterials 12(1), 18 (2021).
https://doi.org/10.3390/jfb12010018
|
| [23] |
Alizadeh-Osgouei, M., Li, Y., Wen, C.: A comprehensive review of biodegradable synthetic polymer-ceramic composites and their manufacture for biomedical applications. Bioactive Materials 4(1), 22-36 (2019).
https://doi.org/10.1016/j.bioactmat.2018.11.003
|
| [24] |
Jiménez-Pérez, A., Martínez-Alonso, M., García-Tojal, J.: Hybrid Hydroxyapatite-Metal Complex Materials Derived from Amino Acids and Nucleobases. Molecules 29(18), 4479 (2024).
https://doi.org/10.3390/molecules29184479
|
| [25] |
Predoi, D., Ciobanu, S. C., Iconaru, S. L., Ţălu, Ş., Ghegoiu, L., Matos, R. S., da Fonseca Filho, H. D., Trusca, R.: New Physico-Chemical Analysis of Magnesium-Doped Hydroxyapatite in Dextran Matrix Nanocomposites. Polymers 16(1), 125 (2024).
https://doi.org/10.3390/polym16010125
|
| [26] |
Geuli, O., Metoki, N., Zada, T., Reches, M., Eliaz, N., Mandler, D.: Synthesis, coating, and drug-release of hydroxyapatite nanoparticles loaded with antibiotics. Journal of Materials Chemistry B 5, 7819-7830 (2017).
https://doi.org/10.1039/C7TB02105D
|
| [27] |
Chung, J. H., Kim, Y. K., Kim, K. H., Kwon, T. Y., Vaezmomeni, S. Z., Samiei, M., Akbarzadeh, A.: Synthesis, characterization, biocompatibility of hydroxyapatite-natural polymers nanocomposites for dentistry applications. Artificial Cells, Nanomedicine, and Biotechnology 44(1), 277-284 (2016).
https://doi.org/10.3109/21691401.2014.944644
|
| [28] |
Hassanzadeh, A., Ashrafihelan, J., Salehi, R., Rahbarghazi, R., Firouzamandi, M., Ahmadi, M., Aghazadeh, M.: Development and biocompatibility of the injectable collagen/nano-hydroxyapatite scaffolds as in situ forming hydrogel for the hard tissue engineering application. Artificial Cells, Nanomedicine, and Biotechnology 49(1), 136-146 (2021).
https://doi.org/10.1080/21691401.2021.1877153
|
Cite This Article
-
APA Style
Faye, A., Dieng, S. M., Ciss, M. J. A., Djiboune, A. R., Souho, B., et al. (2026). Physicochemical Development and Characterization of a Hydroxyapatite-Biocompatible Polymer Matrix for Controlled Insulin Release. European Journal of Biophysics, 14(1), 14-23. https://doi.org/10.11648/j.ejb.20261401.13
 Copy
|
Copy
|
 Download
Download
ACS Style
Faye, A.; Dieng, S. M.; Ciss, M. J. A.; Djiboune, A. R.; Souho, B., et al. Physicochemical Development and Characterization of a Hydroxyapatite-Biocompatible Polymer Matrix for Controlled Insulin Release. Eur. J. Biophys. 2026, 14(1), 14-23. doi: 10.11648/j.ejb.20261401.13
 Copy
|
Copy
|
 Download
Download
AMA Style
Faye A, Dieng SM, Ciss MJA, Djiboune AR, Souho B, et al. Physicochemical Development and Characterization of a Hydroxyapatite-Biocompatible Polymer Matrix for Controlled Insulin Release. Eur J Biophys. 2026;14(1):14-23. doi: 10.11648/j.ejb.20261401.13
 Copy
|
Copy
|
 Download
Download
-
@article{10.11648/j.ejb.20261401.13,
author = {Abdou Faye and Sidy Mouhamed Dieng and Marie Jeanne Arlette Ciss and Alphonse Rodrigue Djiboune and Benedicte Souho and Mamadou Soumboundou and Papa Mady Sy and Gora Mbaye and Mounibe Diarra and Louis Augustin Diaga Diouf},
title = {Physicochemical Development and Characterization of a Hydroxyapatite-Biocompatible Polymer Matrix for Controlled Insulin Release},
journal = {European Journal of Biophysics},
volume = {14},
number = {1},
pages = {14-23},
doi = {10.11648/j.ejb.20261401.13},
url = {https://doi.org/10.11648/j.ejb.20261401.13},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.ejb.20261401.13},
abstract = {This study investigates the controlled release of insulin from composite matrices based on hydroxyapatite and Eudragit® polymers, with the aim of developing an efficient and tunable oral or implantable insulin delivery system. Hydroxyapatite was synthesized by chemical precipitation method with a Ca/P molar ratio of 1.66, closely mimicking the stoichiometry of biological apatite, and subsequently thermally treated at 800°C to enhance its crystallinity, mechanical strength, and structural stability. Two distinct matrix formulations were developed using Eudragit® RSPM and Eudragit® RS100, two pH-independent, water-insoluble polymers known for their controlled permeability properties, and were thoroughly characterized in terms of their technological and pharmacotechnical properties. Evaluation demonstrated good matrix cohesion, adequate hardness, and full compliance with mass uniformity requirements according to pharmacopoeial standards. In vitro release studies conducted under physiologically relevant conditions revealed clearly differentiated kinetic profiles depending on both the formulation type and the insulin loading dose. The first batch exhibited a classical first-order release kinetics, achieving complete insulin release over an extended period of seven days, suggesting a diffusion-controlled mechanism. In contrast, the second batch displayed a sigmoidal release profile accurately described by the Boltzmann equation (r² = 0.99), characterized by a marked acceleration phase beginning on the third day, indicative of a more complex release mechanism. These findings demonstrate that insulin loading significantly influences the release mechanism and overall kinetics, highlighting the strong potential of hydroxyapatite-polymer composite matrices for the rational design of advanced controlled insulin delivery systems.},
year = {2026}
}
 Copy
|
Copy
|
 Download
Download
-
TY - JOUR
T1 - Physicochemical Development and Characterization of a Hydroxyapatite-Biocompatible Polymer Matrix for Controlled Insulin Release
AU - Abdou Faye
AU - Sidy Mouhamed Dieng
AU - Marie Jeanne Arlette Ciss
AU - Alphonse Rodrigue Djiboune
AU - Benedicte Souho
AU - Mamadou Soumboundou
AU - Papa Mady Sy
AU - Gora Mbaye
AU - Mounibe Diarra
AU - Louis Augustin Diaga Diouf
Y1 - 2026/03/10
PY - 2026
N1 - https://doi.org/10.11648/j.ejb.20261401.13
DO - 10.11648/j.ejb.20261401.13
T2 - European Journal of Biophysics
JF - European Journal of Biophysics
JO - European Journal of Biophysics
SP - 14
EP - 23
PB - Science Publishing Group
SN - 2329-1737
UR - https://doi.org/10.11648/j.ejb.20261401.13
AB - This study investigates the controlled release of insulin from composite matrices based on hydroxyapatite and Eudragit® polymers, with the aim of developing an efficient and tunable oral or implantable insulin delivery system. Hydroxyapatite was synthesized by chemical precipitation method with a Ca/P molar ratio of 1.66, closely mimicking the stoichiometry of biological apatite, and subsequently thermally treated at 800°C to enhance its crystallinity, mechanical strength, and structural stability. Two distinct matrix formulations were developed using Eudragit® RSPM and Eudragit® RS100, two pH-independent, water-insoluble polymers known for their controlled permeability properties, and were thoroughly characterized in terms of their technological and pharmacotechnical properties. Evaluation demonstrated good matrix cohesion, adequate hardness, and full compliance with mass uniformity requirements according to pharmacopoeial standards. In vitro release studies conducted under physiologically relevant conditions revealed clearly differentiated kinetic profiles depending on both the formulation type and the insulin loading dose. The first batch exhibited a classical first-order release kinetics, achieving complete insulin release over an extended period of seven days, suggesting a diffusion-controlled mechanism. In contrast, the second batch displayed a sigmoidal release profile accurately described by the Boltzmann equation (r² = 0.99), characterized by a marked acceleration phase beginning on the third day, indicative of a more complex release mechanism. These findings demonstrate that insulin loading significantly influences the release mechanism and overall kinetics, highlighting the strong potential of hydroxyapatite-polymer composite matrices for the rational design of advanced controlled insulin delivery systems.
VL - 14
IS - 1
ER -
 Copy
|
Copy
|
 Download
Download
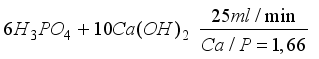 >
>